
Clean hydrogen production, directly from sunlight and water
In the future, solar hydrogen farms will enable clean hydrogen production directly from sunlight and water, whenever the sun is shining. On every panel, the reaction becomes visible as thousands of hydrogen bubbles travel inside the panel — a simple, natural sign of fuel being created from light.
We collect these hydrogen bubbles safely within the system and store the gas on-site. This allows industries, communities, mobility hubs, and remote locations to generate their own clean fuel locally, without relying on centralized infrastructure or long-distance transport.
The system scales naturally: add more panels to produce more fuel. Entire fields, rooftops, or industrial areas could become decentralized hydrogen farms.
This is our future product vision — accessible, modular clean-fuel production for a wide range of users. Our goal is to make hydrogen generation as familiar and widespread as solar electricity is today, giving societies a new way to produce energy directly from the sun.

The membrane is the core of our technology
Which is why we constantly develop their performance features at the lab: efficiency, lifetime, and stability.
Photocatalysts are semiconductors such as used in any technology, but instead of only conducting electricity they are responsive to solar light. And with these particles we are able to transform solar energy directly to hydrogen gas. This direct conversion from sunlight to fuel is what allows our panels to operate without any external electricity, and makes the system fundamentally simpler than conventional electrolysis-based hydrogen production.
Our membranes are engineered in a way that this process happens efficiently under visible light, not just under ultraviolet light as in early photocatalysis research from the 1970s. By designing materials that respond to the broad solar spectrum and integrating them into a stable membrane architecture, we enable practical, scalable, and electricity-free hydrogen production.
At the molecular level
Our technology uses photocatalytic water splitting — one of the simplest conceptual routes for producing clean hydrogen, but also one of the most technically demanding. The principle is often called artificial photosynthesis because, like plants, our materials use sunlight to drive a chemical reaction. In our case, the reaction is the splitting of water into hydrogen and oxygen in following process.
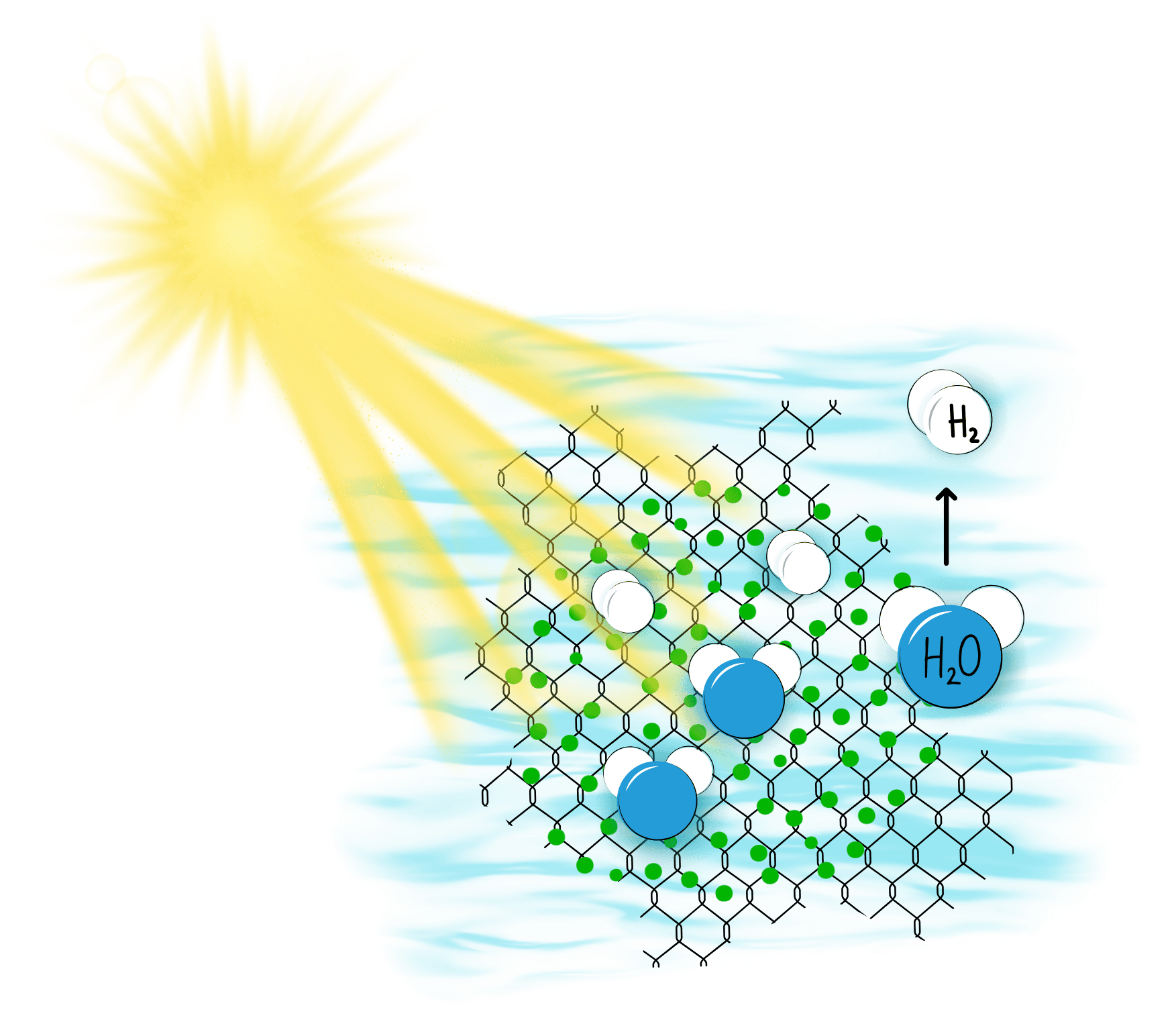
- Light absorption
Our semiconductor photocatalysts absorb visible sunlight. This energy excites electrons inside the material, creating electron–hole pairs. - Charge separation
The electrons and holes move to different regions of the material. This separation is crucial — it prevents the charges from recombining and allows them to perform useful chemistry. - Water splitting reactions
Once separated, these charges drive the two half-reactions of water splitting:
– Hydrogen Evolution Reaction (HER): electrons reduce water to form hydrogen gas (H₂).
– Oxygen Evolution Reaction (OER): holes oxidize water to release oxygen (O₂).
Together, these two reactions convert sunlight and water directly into hydrogen fuel.

